Mark Chandy

Scientist
MD, Ph.D., FRCPC
Why I Became a Scientist
I have an extensive family history of diabetes and cardiovascular disease. All my relatives have diabetes, and all the males have premature coronary disease except for my father, whom I convinced not to smoke. Naturally, with this striking family history of cardiometabolic disease, I am motivated to change the scope and dimension of cardiovascular disease diagnosis and management. Specifically, I want to understand why some individuals are more prone to cardiovascular disease and whether genes or environment or perhaps a combination of both are the major drivers.
Research Summary
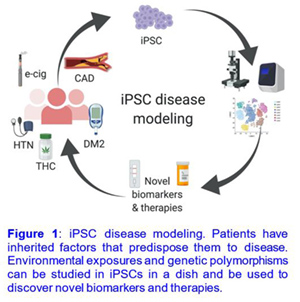 My research employs molecular biology to understand the pathophysiology of cardiovascular disease using human induced pluripotent stem cells (iPSC)-derived tissue. Advances in next-generation sequencing (NGS), proteomics, and gene editing have made it possible to decipher the mechanisms of cardiovascular disease.
My research employs molecular biology to understand the pathophysiology of cardiovascular disease using human induced pluripotent stem cells (iPSC)-derived tissue. Advances in next-generation sequencing (NGS), proteomics, and gene editing have made it possible to decipher the mechanisms of cardiovascular disease.
Research Questions
How does the environment affect the cardiovascular system?
Induced pluripotent stem cells (iPSCs) are a powerful model system for medical discovery and translational research. At present, most studies have focused on the potential for iPSCs for regenerative medicine, drug discovery, and disease modeling. However, iPSCs are also a powerful model system to investigate the effects of environmental risk factors such as e-cigarettes, marijuana, air pollution, and opioids on the cardiovascular system.
How does type 2 diabetes and insulin resistance exacerbate cardiovascular disease?
Insulin resistance is central to the development of type 2 diabetes (T2D) and presents a major risk factor for cardiovascular disease. Insulin resistance is difficult to diagnose before the onset of T2D and is often underdiagnosed with the prevalence of insulin resistance estimated to be three times greater than T2D. The causes of insulin resistance and how it impacts the vascular system are not fully understood. Therefore, there is a need to identify early diagnostics for insulin resistance, as well as potential therapeutic targets.
How can iPSC-disease modeling be used to discover novel biomarkers and treatments for cardiovascular disease?
Cells can communicate with each other via small vesicle packets called exosomes. Exosomes contain nucleic acid, protein, and lipids. The collection of molecules released by secreted exosomes can influence nearby cells’ behavior, function, development, and even identity. Exosomes and their content might be implicated in the mechanisms of cardiomyopathy and could serve as biomarkers or targets for drug discovery.
Education
- 2000 – 2008 MD and Ph.D. Pennsylvania State University, USA
- 1998 – 2000 M.Sc. University of Toronto, Toronto, Canada
- 1994 – 1998 B.Sc. University of Toronto, Toronto, Canada
Postgraduate Medical and Research Training
- 2016 – 2021 Postdoctoral Research Fellowship in Stem Cell Biology, Cardiovascular Research Institute, Stanford University, USA
- 2014 – 2016 Postdoctoral Research Fellowship in Translational Biology, Toronto General Research Institute, University of Toronto, Toronto, Canada
- 2011 – 2014 Adult Cardiology, University of Toronto, Toronto, Canada
- 2008 – 2011 Internal Medicine, University of British Columbia, Vancouver, Canada
Awards
- 2014 – 2019 Clinician-Scientist Training Program, University of Toronto
- 2016 – 2018 Detweiler Traveling Fellowship
- 2000 – 2008 Medical Scientist Training Program
Publications
Contact Info
Email: mark.chandy@lhsc.on.ca
Website: www.chandylab.org








