Hyperpolarized Metabolic Probes
Seminal research by K. Golman et al. has demonstrated the use of hyperpolarized substances with enriched 13C content as novel contrast agents for a wide range of MRI applications. Research underway at the Robarts Research Institute will use hyperpolarized endogenous compounds to map cellular metabolism with a primary focus on pre-clinical cancer imaging research.
Generally speaking 13C nuclei are not imaged with MRI in clinical practice or even for in vivo research because, under normal imaging circumstances, they are not detectable. Hyperpolarization is the ability to increase the magnetization of a compound in vitro. The enhancement of the magnetization can be a factor of nearly 100,000 permitting spectroscopic imaging of these compounds after injection into an animal model. Recent research has identified over 70 possible 13C-enriched endogenous compounds that are involved in cellular metabolism that meet a long list of criteria to be both safe for in vivo use and possess all the physical attributes necessary for hyperpolarization and subsequent imaging.
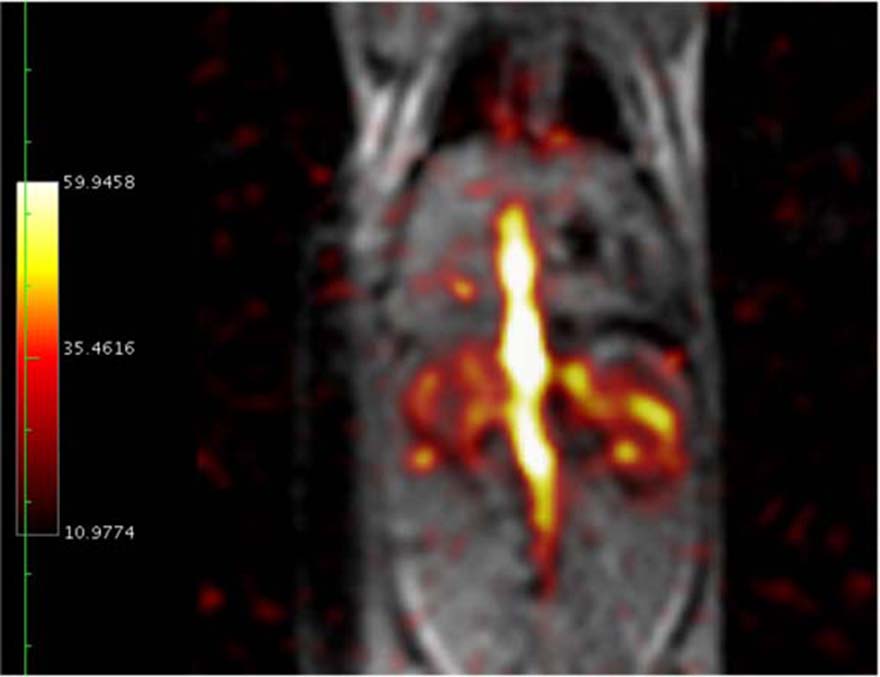
Using magnetic resonance spectroscopic imaging, these injected hyperpolarized endogenous compounds and their metabolites can be spatially and temporally imaged. Thus it is possible to use this technology to differentiate cellular metabolism for healthy and diseased tissue in an animal model of cancer. As a particular example using in vivo imaging, it has been demonstrated that the ratio of pyruvate to its metabolite lactate can be used as a biomarker to distinguish cancerous tissue for a variety of different cancer cells.
Another application of this method is in vivo imaging of hyperpolarized 13C-enriched bicarbonate in equilibrium with carbon dioxide. Magnetic resonance spectroscopic imaging of these two compounds can produce very accurate and detailed maps of tissue pH. The sensitivity of this cellular imaging technique may provide a means of early detection of inflammatory events at the cellular level that may not be detected by traditional methods.
Our Research Objective
The research undertaken by this group has a primary focus on cancer imaging using hyperpolarized endogenous MR probes such as to look at:
- Detection limits for cancer imaging,
- Characterization of tumour and tissue, and
- Monitoring tumor response to treatment and
- Inflammatory response quantification using tissue pH mapping.
This research will be conducted in collaboration with other Robarts scientists - primarily Dr. Paula Foster who leads the program in Cellular and Micro Imaging at the Robarts Research Institute. This is an interdisciplinary research program focused on the development and applications of pre-clinical micro-imaging methods for studying disease.
Dr. Foster has an interest in imaging the evolution of inflammation to study diseases such as organ rejection, demyelination, infection and spinal cord injury. We also will work closely with Dr. Giles Santyr to study radiation induced lung injury using a rodent model. This unique collaboration will combine our abilities of our group to image 13C-enriched substrates with the expertise of the Santryr lab in pulmonary imaging using hyperpolarized 129Xe gas.
Our Contribution
We have amassed a new laboratory immediately adjacent to the General Electric Healthcare MR750 3T imager in the 2nd-floor Imaging Centre at the Robarts Research Institute capable of hyperpolarization of isotopically enriched substrates. We also have furnished a dedicated RF laboratory to develop all the specialized hardware for detection of 13C nuclei including a dual-tuned 1H - 13C birdcage RF coil for rodents and the first ever eight-channel receive array. We have integrated this RF hardware with our existing high-performance gradient inserts to develop a novel imaging platform capable of both high-resolution proton imaging and rapid spectroscopic imaging of hyperpolarized substrates.
Key Questions
- How sensitive is this technique for early cancer detection?
- Can biomarkers based on hyperpolarized endogenous compounds be found to predict or measure a cancer cell's ability to proliferate or metastasize?
- How early can this technique be used to predict therapeutic response for cancer treatment?
- How sensitive is this method for detecting early inflammatory response?








